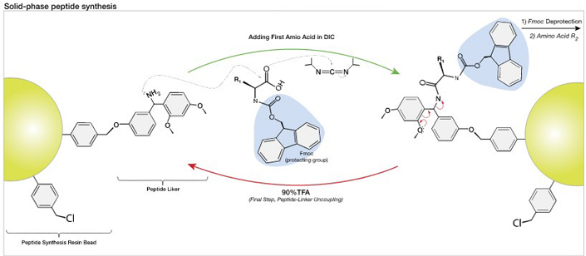
SPPS uses an inert and insoluble resin (or carrier beads) to serve as a template upon which a growing chain of amino acids may be attached. Once the desired amino acid chain or peptide is built, the peptide is cleaved from the resin by a selective reaction. The most significant advantage of solid phase over "traditional" solution phase chemistry is that following each cycle of amino acid addition to the chain, the resin (and its attached peptide) remains insoluble and excess reagents and unreacted materials can be easily rinsed away. Thus, step-by-step purification is conducted through simple washing of the "caviar-like" resin rather than using chromatographic means.
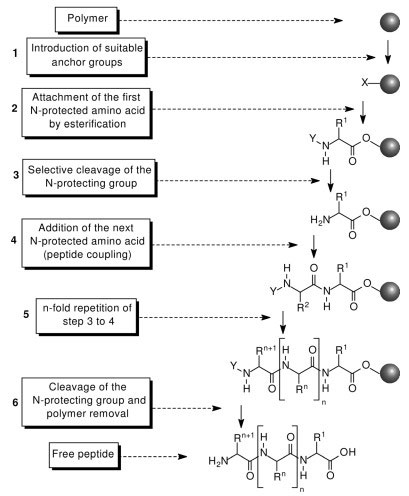
There are two major forms of solid phase peptide synthesis – (1) Fmoc (base labile alpha-amino protecting group) and (2) t-Boc (acid labile protecting group). Each method involves different resins and amino acid side-chain protection and consequent cleavage/deprotection steps. Fmoc chemistry is known for generating peptides of higher quality and in greater yield than t-Boc chemistry. Impurities in t-Boc-synthesized peptides are mostly attributed to cleavage problems, dehydration and t-butylation. After cleavage from the resin, peptides are usually purified by reverse phase HPLC using columns such as C-18, C-8, and C-4.
